 BDR proteins
BDR proteins
A family of negative transcription elongation factors protects the genome of Arabidopsis thaliana from transcriptional interferences
Abstract
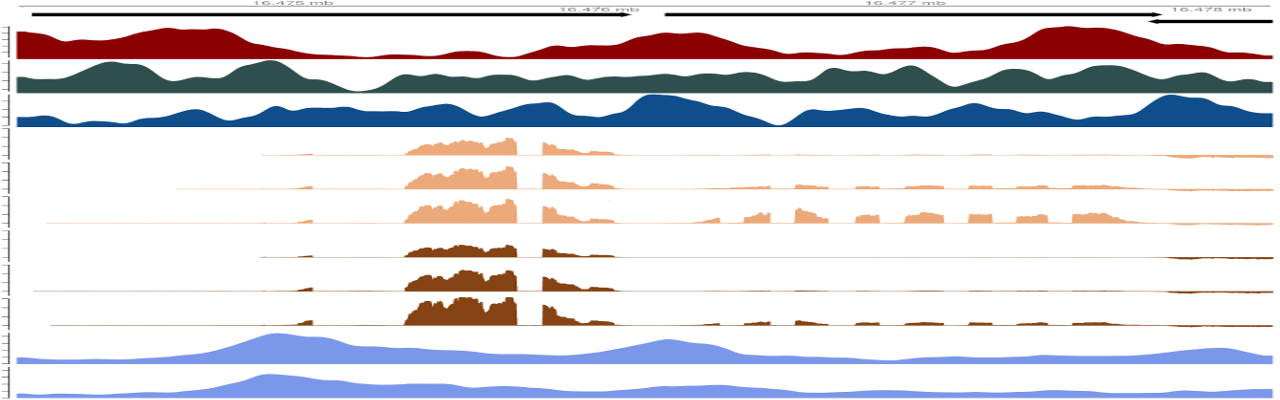
During the last decade, much progress has been made in our understanding of how transcription elongation is regulated and how it affects gene expression, RNA maturation and chromatin organization. Like nematodes and yeast, plants lack the negative elongation factor (NELF) and do not exhibit widespread promoter-proximal pausing of RNA polymerase II (RNAPII) after transcription initiation. In this work, we characterized the genome-wide function of a new family of negative transcription elongation factors in Arabidopsis. We named the 3 members BorDeRs (BDR1 2 and 3) because BDR1 and BDR2 are enriched, together with RNAPII, at evolutionarily-conserved, nucleosome-depleted regions located at the borders of a large fraction of genes across the genome. BDR1 also interacts with FPA, a factor involved in RNA 3' processing and transcription termination. RNA-seq defined repertoires of hundreds of genes that were either up- or downregulated following the disruption of all three BDR genes in Arabidopsis. By integrating genome-wide data on BDRs, RNAPII and histone modifications, a picture is emerging in which BDRs cooperate with transcription factors such as WRKY to regulate the expression of genes involved in plant defense responses. Unexpectedly, we found that, in bdr triple mutant plants, a large fraction of genes appeared downregulated as a result of transcriptional interferences. In these mutants, defective transcriptional termination and 3' RNAPII pausing at the upstream tandem gene appear to generate cis interference with transcriptional initiation at the promoter of downregulated downstream genes. Altogether, our data illustrate how BDR negative elongation factors have opposite effects on gene expression by influencing different steps of the transcription cycle and strikingly play a crucial role in limiting transcriptional interferences at closely-spaced tandem genes.